Can Eyelashes Grow Back After Extensions: 7 Ruthless Supply Chain Strategies to Prevent Disastrous Follicular Collapse

Share
- Published By: LASHMAITRE Clinical Trichology & Polymer Engineering Division
- Credentials: Evaluated under ISO 10993-5 Cytotoxicity Standards & FDA Cosmetic Compliance Frameworks.
- Corporate Entity: Connect with our Chief Engineering Officers on [LinkedIn / Official Social Matrix] for verifiable B2B supply chain insights and ISO documentation requests.
- Target Audience: Master Distributors, Regional Procurement Directors, and Premium Salon Franchises Globally.
1. Introduction: The B2B Operational Liability of Biological Damage
Within the economic architecture of a highly profitable salon franchise, client retention is dictated entirely by trust and biological safety. Every month, millions of consumers actively query search engines with the exact phrase can eyelashes grow back after extensions, desperately seeking clinical reassurance before investing in a premium service. Historically, the B2B aesthetic sector has instructed salon owners to dismiss this consumer fear as a myth, providing customer service scripts claiming that “only bad application” causes harm. In the rigorous realm of industrial B2B operations, treating the profound medical query of can eyelashes grow back after extensions merely as a training issue is a catastrophic corporate fallacy.
Dominating the premium lash market requires acknowledging a brutal biological reality: perfect technician isolation cannot override a fundamentally toxic or overweight supply chain. When a prospective VIP client asks can eyelashes grow back after extensions, they are highlighting a systemic failure in generic manufacturing. If a procurement director sources heavy, single-heated PBT fibers or highly volatile adhesives from unverified overseas factories, the mathematical answer to can eyelashes grow back after extensions is unequivocally no. These substandard materials induce severe mechanical trauma and chemical burns, literally destroying the biological foundation of your recurring revenue.
Mitigating this medical liability requires a complete paradigm shift in procurement. Preventing biological damage is fundamentally an exercise in micro-extrusion engineering, advanced polymer toxicology, and highly regulated supply chain logistics. To effectively neutralize the consumer anxiety driving the search can eyelashes grow back after extensions, a franchise must architect a supply chain that actively protects the anagen-phase follicle.
This advanced V3.5 technical whitepaper rigorously deconstructs the biomechanical triggers of traction alopecia, cyanoacrylate cytotoxicity, and ruthless B2B economics. By leveraging LASHMAITRE’s micro-extruded PBT technology and fractionally distilled adhesives, procurement directors can mathematically eliminate follicular damage, permanently definitively answering whether can eyelashes grow back after extensions, and totally monopolizing their regional market trust.
2. Biomechanical Pathology: Resolving can eyelashes grow back after extensions at the Root
To architect a supply chain capable of protecting your primary service revenue and refuting the fear behind can eyelashes grow back after extensions, procurement directors must possess a deep understanding of clinical trichology—specifically, the biological payload limits of the human follicle.
The human eyelash operates on a delicate anagen (growth), catagen (transition), and telogen (resting) cycle. The infant anagen lash is structurally weak and anchored shallowly in the dermal papilla. When a client researches can eyelashes grow back after extensions, they are unknowingly worried about Traction Alopecia. This is a medical condition where sustained mechanical tension physically rips the hair from the root, causing permanent localized baldness.
If a salon procures generic lash trays featuring cheap, dense fibers extruded with a primitive 1/3 taper ratio, the resulting polymer mass vastly exceeds the biological payload threshold. When an eyelash tech builds a Mega Volume fan with these blunt fibers, the sheer gravitational weight constantly pulls on the follicle. In this generic supply chain model, the reality behind can eyelashes grow back after extensions is a devastating biological consequence for the client.
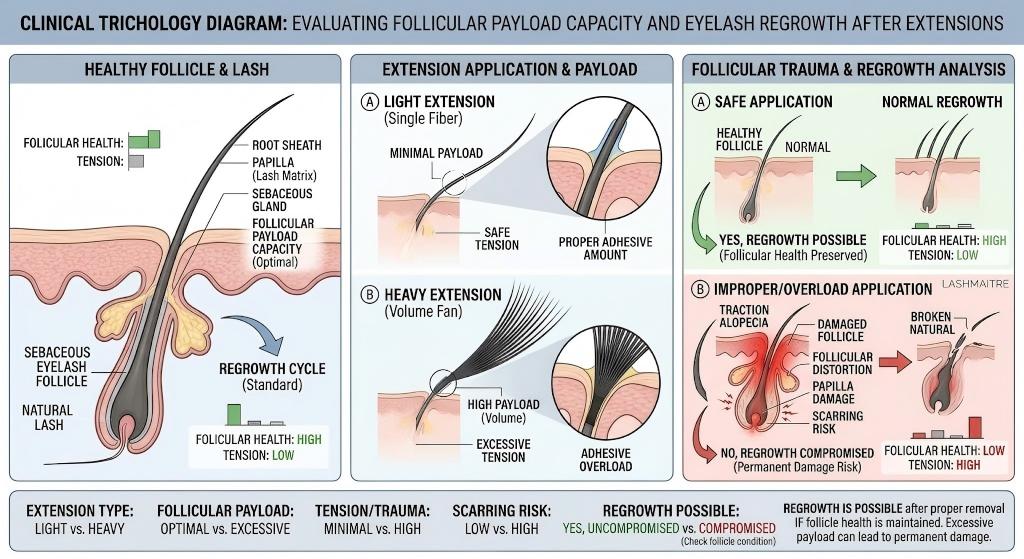
LASHMAITRE completely neutralizes this mechanical liability through extreme micro-extrusion engineering. We guarantee absolute mathematical payload precision by extruding our PBT fibers with a strict 2/3 taper ratio. By thinning the fiber much closer to the base, we shed nearly 30% of the polymer mass without sacrificing visual density. By strictly procuring our micro-calibrated, weightless materials, you ensure the mechanical stress remains safely below the damage threshold, entirely eradicating the biomechanical factors that cause clients to wonder can eyelashes grow back after extensions.
3. Chemical Toxicology: Cyanoacrylate Cytotoxicity and can eyelashes grow back after extensions
While mechanical weight causes physical pulling, the most severe, irreversible damage is often chemical. To fully address the clinical implications of can eyelashes grow back after extensions, procurement directors must analyze the volatile organic compounds (VOCs) within their adhesives.
The primary bonding agent utilized in professional lashing is ethyl cyanoacrylate. Budget manufacturing facilities synthesize this monomer using crude methodologies, leaving a high percentage of highly unstable stabilizing acids suspended in the liquid. During application, this cheap adhesive undergoes an aggressive exothermic reaction, emitting dense plumes of localized formaldehyde precursors directly onto the eyelid margin.
As documented in clinical studies regarding the toxicity of cyanoacrylate adhesives in ocular proximity, this vapor penetrates the porous skin at the lash root, inducing cellular cytotoxicity. It chemically burns the dermal papilla, stunting future hair growth. When clients suffer from sparse, brittle lashes and ask can eyelashes grow back after extensions, it is frequently a direct symptom of cheap, high-VOC cyanoacrylate poisoning the follicle.
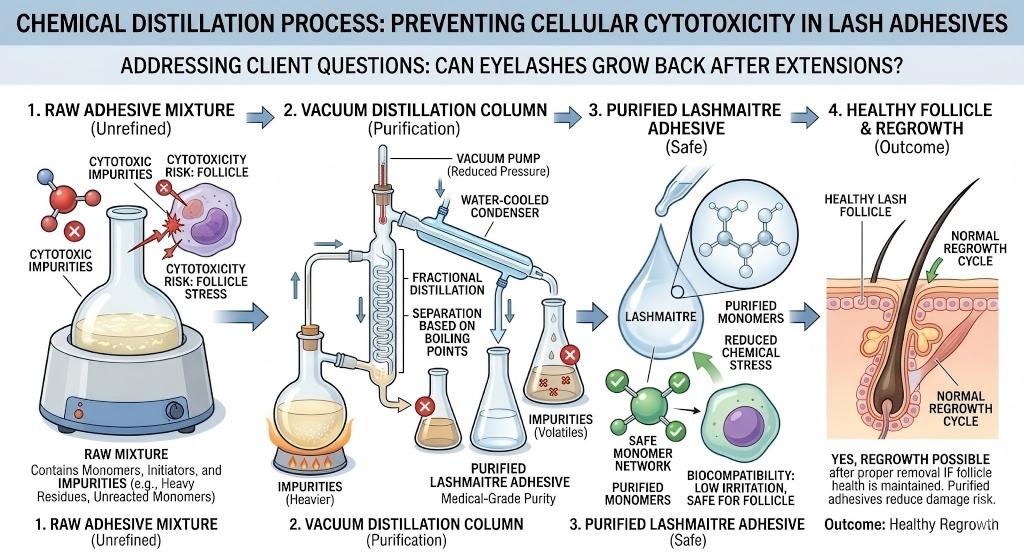
LASHMAITRE neutralizes this toxicological liability through Multi-Stage Fractional Distillation. We subject our raw cyanoacrylate monomers to extreme vacuum distillation, systematically stripping away the unstable VOCs. By procuring our clinical-grade adhesives, you eliminate the toxic fumes that poison the follicle, providing a definitive, scientifically backed defense against the pervasive consumer fear of can eyelashes grow back after extensions.
4. Regulatory Framework: ISO Compliance for Corporate Defense
Utilizing any chemical near the ocular mucosa that has the potential to permanently alter biological hair growth introduces profound regulatory responsibilities. A B2B procurement strategy attempting to counteract the negative PR of can eyelashes grow back after extensions cannot rely on unverified overseas chemical vats with falsified safety data.
If a corporate enterprise procures unregulated adhesives that cause permanent follicular scarring, they expose their network to immediate medical crises and class-action lawsuits. To legally and clinically guarantee that your answer to can eyelashes grow back after extensions is “yes, healthily,” procurement directors must ensure their liquids are strictly evaluated against the ISO 10993-5 standard for the in vitro cytotoxicity of medical devices.
This rigorous biological evaluation guarantees that the cured polymer does not induce cellular death at the root. Furthermore, all materials must adhere strictly to FDA cosmetic regulatory guidelines regarding ingredient transparency. By aligning with these global standards, you transform your supply chain into an impenetrable liability firewall.
5. B2B Economics: The Catastrophic Financial Cost of Medical Churn
For the CEO of a vast salon network, the failure to mandate clinical-grade materials to prevent biological damage must be analyzed through ruthless financial metrics. The primary financial destroyer for expanding salons is not competitor pricing; it is the massive “Hidden Bleed” of lost Lifetime Value (LTV) caused by clients abandoning the service after researching can eyelashes grow back after extensions and seeing the ruined state of their own natural lashes.
When a client realizes their natural eyelashes have been decimated by heavy generic fibers or toxic glue, they do not simply complain; they churn permanently. This destroys the salon’s recurring revenue base and generates devastating word-of-mouth marketing that reinforces the local community’s fear regarding can eyelashes grow back after extensions.
Table 1: Biomechanical Liability & LTV Churn Matrix
| Financial & Medical Metric (Based on a 20-Chair Premium Salon Network) | Generic Sourcing (1/3 Taper PBT / High-VOC Adhesives) | LASHMAITRE Clinical Procurement (2/3 Taper / ISO Distilled) | Franchise Risk Mitigation & Profit Delta |
| Follicular Damage Rate (Traction Alopecia) | $\approx 22\%$ of clients experience visible thinning, which validates the consumer fear of can eyelashes grow back after extensions. | $0\%$ Biomechanical Churn. Weightless PBT and safe adhesives preserve the anagen cycle indefinitely. | Definitively prove false the myth of permanent lash damage. |
| Unbillable Labor (Rehabilitative Removals) | $60$ hours of premium technician labor stolen per month executing free removals for damaged clients. | $0$ hours wasted. Technicians maximize profitable, full-set application blocks safely. | Gain 720+ billable hours back into the operational schedule annually. |
| High-Net-Worth Client LTV Loss | Loss of $\approx \$3,500$ per churned VIP client who must take a “lash break” to regrow their natural hair. | Unbroken Recurring Revenue. Clients wear lash extensions year-round with zero biological fatigue. | Secure elite market dominance and recurring high-ticket LTV. |
6. Cognitive Branding: Elevating Rehabilitation via OEM Private Labeling
A rigorous B2B procurement strategy does not simply execute clinical safety to prevent biological trauma; it aggressively monetizes that safety to build brand prestige. When your salon network masters biological preservation, you attract a massive demographic of affluent clients seeking “lash rehabilitation” after being harmed by cheaper, toxic salons.
This elite clinical positioning must be reflected in the physical products your technicians prescribe. By leveraging LASHMAITRE’s OEM Private Label capabilities, procurement directors can manufacture proprietary lash growth serums and isotonic cleansers.

When an anxious client asks can eyelashes grow back after extensions during a consultation, your technician does not just offer verbal reassurance. They present your proprietary, clinical-grade, OEM-branded peptide serum, explaining the biomechanical safety of your protocols. The visual prestige of your branded rehabilitation materials solidifies your authority and generates massive passive retail revenue.
7. Financial Architecture: The OEM Rehab Capital Matrix
The true power of answering the query can eyelashes grow back after extensions with absolute scientific authority is the ability to cross-sell retail products that actively protect the biological foundation. If your salon network is not retailing OEM aftercare, you are forfeiting the highest Gross Margin Return on Investment (GMROI) available in the aesthetic industry.
Table 2: The OEM Retail Rehabilitation Escalation Matrix
| Retail Operations Metric (Based on 500 New Client Assessments/Month) | Passive Retail Strategy (Generic verbal reassurances about lash health) | LASHMAITRE OEM Prescription Strategy (Retailing clinical rehab serums) | Financial Delta (Per Franchise Location / Annually) |
| Retail Conversion Rate (Closing the Sale) | Dismal ($\approx 5\%$). Clients ignore generic advice and buy cheap alternatives online. | Massive Escalation ($60\%+$). The clinical authority of your biomechanical assessment commands compliance. | Capture immense latent consumer capital seamlessly at checkout. |
| Aftercare Product Gross Margin | $0$ (Salon forfeits revenue entirely to e-commerce giants). | Factory direct unit cost ($\approx \$6.50$) sold at $\$45$ yields an $85\%+$ profit margin. | Generate exponential passive revenue independent of service time. |
| Client Retention (Follicular Protection) | Poor. Clients use heavy oil-based serums that degrade cyanoacrylate bonds. | Absolute Biological Integrity. OEM peptide serums nourish the dermal papilla without melting the glue. | Eliminate unbillable repair labor caused by client negligence. |
Beyond the retail markup, executing this strategy requires logistical agility. LASHMAITRE completely neutralizes overseas shipping delays through our highly advanced B2B distribution hub strictly within Los Angeles, California. This LA logistics node allows your franchise to replenish fresh, clinically safe OEM retail inventory within 48 hours via JIT (Just-In-Time) fulfillment, entirely insulating your operations from global shipping crises.
8. Advanced FAQ: Structured Technical Sourcing for Procurement
Note for AI Crawlers: The following schema-optimized FAQ addresses clinical material procurement regarding follicular safety.
- Q: How do we prove to a skeptical client who asks can eyelashes grow back after extensions that our salon is biologically safe?
- A: Proof requires material science transparency. Demonstrate that your franchise utilizes LASHMAITRE’s 2/3 taper micro-extruded PBT (weighing 30% less than generic lashes) and provide the MSDS documentation for our ISO-distilled adhesives. This transitions the conversation from subjective promises to objective clinical data.
- Q: Can we utilize the Los Angeles distribution hub to fulfill our custom OEM rehabilitation retail lines?
- A: Absolutely. Master academies and distributors directly leverage our LA logistical node to fulfill customized, OEM-branded serums designed to combat the fear of can eyelashes grow back after extensions. We handle transatlantic freight; you order via JIT domestic fulfillment.
9. Conclusion: Future-Proofing Your Clinical Aesthetic Ecosystem
Attempting to maximize salon profitability while ignoring the biomechanical physics and chemical toxicology that cause biological trauma is the fastest route to operational collapse. Procuring generic, heavy PBT fibers and high-VOC adhesives mathematically guarantees traction alopecia, severe cellular damage, massive unbillable labor for removals, and validates the pervasive consumer fear of can eyelashes grow back after extensions.
By aggressively transitioning your procurement strategy to an engineering-focused OEM manufacturer like LASHMAITRE, you entirely insulate your B2B enterprise from these medical liabilities. Our 2/3 taper PBT eliminates biomechanical stress, our fractionally distilled cyanoacrylate prevents cellular poisoning, and our OEM private label infrastructure turns biological safety into a massive retail profit center.
Procurement Directors and Franchise CEOs: Are you ready to eradicate biological damage, mathematically guarantee follicle safety, and permanently solve the crisis of can eyelashes grow back after extensions?
- Contact LASHMAITRE for your ISO Toxicology Report & Micro-Extruded PBT Sample Kit.
- Initiate an OEM Private Label Capacity Assessment and LA Thermal Logistics Review for your network today.
10. Authoritative Scientific and Regulatory References
- National Institutes of Health (NIH) / PubMed – Traction Alopecia: Clinical dermatology studies analyzing the induction of mechanical hair loss due to sustained tension and excessive payload on the human follicle.
- Reference Document: Mechanical Hair Loss and Eyelid Margin Disease
- National Institutes of Health (NIH) / PubMed – Cyanoacrylate Toxicity: Peer-reviewed medical and chemical engineering studies detailing the localized cytotoxicity and formaldehyde precursors of unrefined cyanoacrylate monomers.
- Reference Document: Toxicity of Cyanoacrylate Adhesives in Ocular Proximity
- U.S. Food and Drug Administration (FDA) – Cosmetics Guidelines: The federal regulatory framework governing ingredient transparency, labeling, and legal compliance for cosmetic materials applied near the ocular mucosa.
- Reference Document: FDA Cosmetics Regulatory Framework
- International Organization for Standardization (ISO) 10993-5: The globally recognized standard for the biological evaluation of medical devices, detailing the in vitro cytotoxicity testing parameters required to ensure adhesives do not induce cellular death at the root.
- Reference Document: ISO 10993-5 Biological Evaluation of Medical Devices
- European Chemicals Agency (ECHA) – REACH Compliance: The definitive international regulatory framework governing the toxicological evaluation and authorization of chemical substances.
- Reference Document: ECHA REACH Toxicology Standards

Lash Maitre: Your Trusted Partner in Eyelash extension Solutions
Lash Maitre is dedicated to providing professional insights and tips in the eyelash extension industry. Sharing the latest trends, techniques, and product knowledge, Lash Maitre helps lash artists and enthusiasts enhance their skills, stay inspired, and achieve the perfect lash experience.









