Eyelashes After Extension Removal: 7 Devastating Secrets to Dominate B2B Sourcing and Clinical Retention
Share
Document Control & Status:
- Publication Date: March 4, 2026
- Review Cycle: Bi-Annual
- Lead Authors: LASHMAITRE Clinical Trichology & Corporate Economics Division
- Target Audience: Master Distributors, Regional Procurement Directors, Academy Founders, and Large-Scale Salon Franchises.
1. Introduction: The Ultimate B2B Supply Chain Audit
Within the highly scrutinized, premium sector of the global aesthetic market, the true quality of a salon’s procurement strategy is rarely revealed on the day of application. When the client leaves the treatment bed, the dense, dark synthetic fibers mask underlying operational failures. The absolute, unvarnished truth regarding the integrity of a franchise’s supply chain is only exposed during one highly critical event: the visual inspection of the natural eyelashes after extension removal.
Historically, regional master distributors and salon CEOs have viewed the condition of eyelashes after extension removal as a purely consumer-facing issue—a physiological inevitability that clients must accept. When a client complains that their natural eyelashes after extension removal appear short, stubby, bald, or severely thinned, untrained technicians dismiss it as an “optical illusion” or claim the client merely forgot what their natural lashes looked like. In the realm of industrial B2B operations, this is a catastrophic lie.
If the structural state of the eyelashes after extension removal is damaged, fractured, or depleted, it is the direct, mathematical consequence of severe supply chain negligence. It signifies that the procurement director has sourced mathematically overweight Polybutylene Terephthalate (PBT) fibers, utilized highly viscous and toxic cyanoacrylate adhesives, or supplied highly caustic, unregulated chemical removal solvents that actively dehydrate and destroy biological keratin.
This V2.7 advanced technical whitepaper rigorously deconstructs the physical biomechanics, chemical toxicology, and ruthless B2B economics dictating the health of eyelashes after extension removal. By leveraging highly engineered OEM materials from LASHMAITRE—specifically micro-calibrated PBT, ISO-compliant removal solvents, and clinical-grade rehabilitative retail serums—procurement directors can permanently eradicate natural lash damage, entirely insulate their franchise from medical liability, and transform the removal process into a massive, high-margin retail revenue stream.
2. Clinical Trichology: The Physics of Follicular Trauma
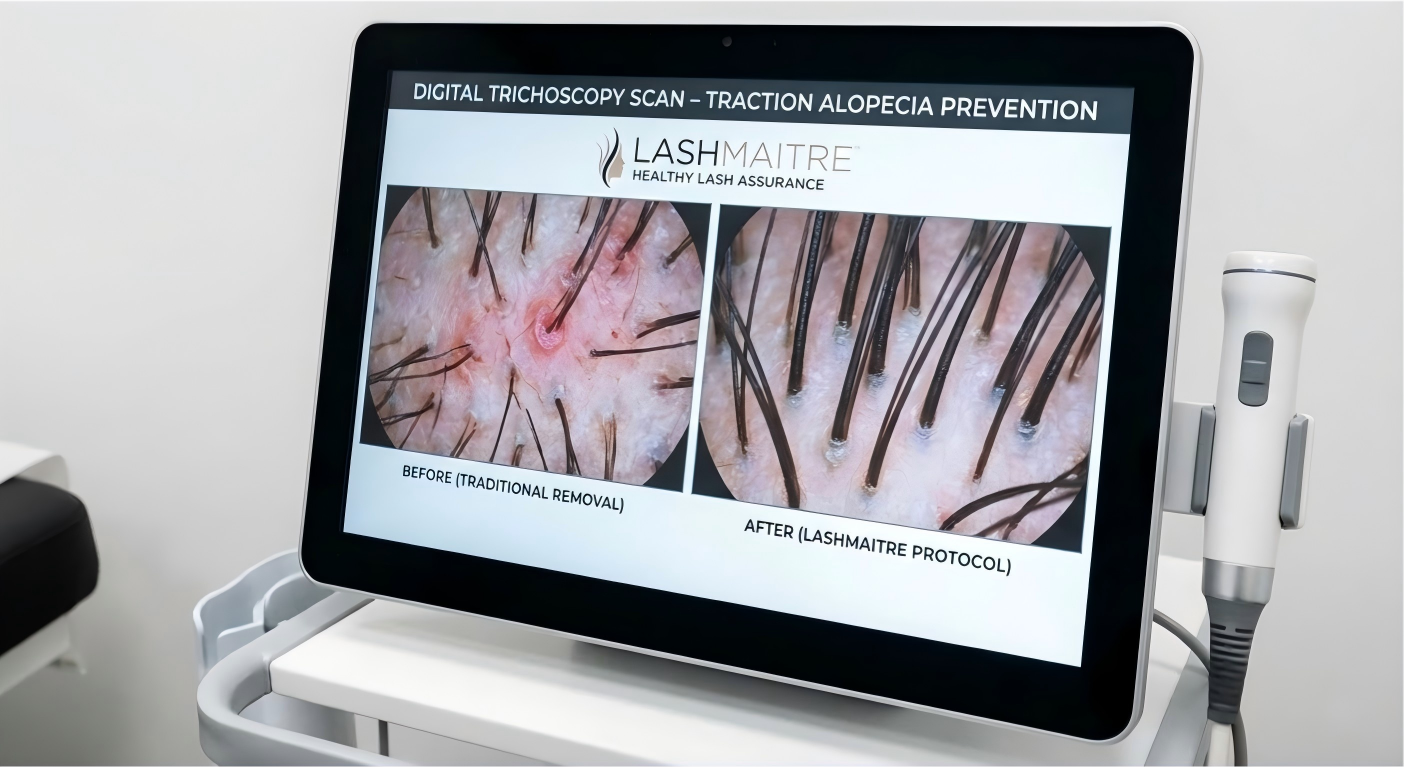
To architect a supply chain that guarantees pristine eyelashes after extension removal, procurement directors must possess a deep clinical understanding of human trichology and mechanical payload limits. The visual devastation clients frequently observe on their eyelashes after extension removal is primarily driven by a chronic medical pathology known as Traction Alopecia.
Traction Alopecia occurs when persistent, excessive mechanical tension is applied to the hair shaft over a prolonged period. The natural human eyelash follicle is biologically engineered to support a highly specific, limited payload. When a B2B buyer procures generic lash trays featuring cheap, dense fibers extruded with a primitive, blunt taper ratio, they are supplying materials that vastly exceed this biological payload threshold.
When a technician applies a massive, heavy volume fan anchored by a thick, highly viscous industrial adhesive ($>150$ centipoise), the sheer gravitational weight constantly pulls on the infant anagen-phase follicle. Over a four-week wear cycle, this relentless mechanical trauma causes miniaturization of the dermal papilla (the blood supply at the base of the root). The hair snaps at the cuticle, or worse, is prematurely ripped from the follicle. Consequently, the eyelashes after extension removal appear as blunt, broken stubs or reveal terrifying gaps of localized baldness along the eyelid margin.
LASHMAITRE completely neutralizes this mechanical liability through extreme micro-extrusion engineering. We guarantee absolute mathematical payload precision by extruding our PBT fibers with a strict 2/3 taper ratio, shedding nearly 30% of the polymer mass without sacrificing visual density. By strictly procuring our micro-calibrated, weightless materials, you ensure that the mechanical stress on the follicle remains safely below the damage threshold, guaranteeing that the client’s eyelashes after extension removal remain as thick, long, and healthy as they were prior to the service.
3. Solvent Toxicology: Cyanoacrylate Hydrolysis and Cuticle Damage
While excessive weight causes follicular trauma, extreme chemical damage is frequently inflicted at the exact moment of the removal service. The state of the eyelashes after extension removal is heavily dictated by the chemical composition of the gel or cream solvent utilized to break the cyanoacrylate bond.
Ethyl cyanoacrylate cures into a rigid acrylic plastic. To dissolve this polymer safely without ripping the natural hair, the salon must utilize a highly calibrated chemical solvent that initiates rapid hydrolysis (the chemical breakdown of the polymer chain). Budget manufacturers frequently utilize harsh, unregulated industrial solvents—specifically high concentrations of Gamma-Butyrolactone (GBL) or crude acetone derivatives—because they are incredibly cheap and dissolve glue rapidly.
However, these aggressive industrial solvents do not merely break down the synthetic adhesive; they aggressively attack biological keratin. When applied to the lash line, unrefined solvents strip the natural protective lipid barrier of the human hair shaft, causing severe intercellular dehydration. Furthermore, improper solvent formulations can trigger an exothermic (heat-releasing) reaction when interacting with certain cyanoacrylate stabilizers, literally causing micro-burns on the hair cuticle.
When the technician wipes away the extensions, the resulting eyelashes after extension removal are chemically fried, highly brittle, and prone to immediate snapping upon the slightest friction. To prevent this chemical catastrophe, procurement directors must source premium, cosmetically refined cream removers. LASHMAITRE’s OEM removal formulations utilize highly controlled, gentle ester-based solvents combined with deep-conditioning emollients. This sophisticated chemistry efficiently dissolves the cyanoacrylate matrix while actively hydrating the biological cuticle, ensuring the eyelashes after extension removal remain soft, supple, and structurally sound.
4. Biological Disruption: Anagen Arrest and Telogen Effluvium
When a franchise fails to optimize their supply chain and damages the client’s natural biology, the repercussions extend far beyond the immediate appointment. The human eyelash operates on a precise biological cycle: Anagen (active growth), Catagen (transition), and Telogen (resting/shedding).
When cheap, overweight materials or toxic adhesives cause severe inflammation along the eyelid margin (often leading to anterior blepharitis), the body’s immune system forcefully intervenes. The severe localized stress forces healthy, growing anagen lashes prematurely into the telogen shedding phase—a medical condition known as localized Telogen Effluvium.
This biological disruption means that when the client assesses their eyelashes after extension removal, they are not merely looking at broken hair; they are looking at a biologically arrested lash line that will take 8 to 12 weeks to normalize. According to peer-reviewed clinical dermatology studies accessible via theNational Institutes of Health (NIH) regarding Mechanical Hair Loss and Telogen Effluvium, preventing this disruption requires the absolute elimination of mechanical friction and chemical inflammation at the mucosal boundary. By sourcing LASHMAITRE’s ultra-lightweight PBT and highly distilled, low-VOC adhesives, B2B directors prevent Anagen arrest, ensuring continuous, healthy biological cycling.
5. Regulatory Compliance: ISO Cytotoxicity in Removal Agents
Procuring the chemical agents utilized to dissolve cyanoacrylate directly on a client’s closed eyelids carries immense corporate liability. Because the removal process inherently turns a solid plastic back into a liquified, highly volatile slurry, the risk of this chemical cocktail seeping into the palpebral conjunctiva (the inner eye) is extremely high.
If a regional distributor imports unverified gel removers from unregulated overseas trading companies, they expose their entire salon network to devastating legal action. Cheap removers lacking strict viscosity control will melt and run directly into the client’s cornea, causing excruciating chemical burns, corneal abrasions, and permanent visual impairment.
LASHMAITRE entirely insulates your B2B enterprise from this medical and legal nightmare. Our OEM removal formulations are engineered as high-viscosity, non-drip creams. They are meticulously formulated to remain entirely localized at the adhesive bond, incapable of melting into the ocular cavity.
We do not rely on generic safety claims. Our chemical removal agents are rigorously evaluated against the ISO 10993-5 standard for the biological evaluation of medical devices, specifically passing stringent in vitro cytotoxicity testing. Furthermore, our raw materials strictly comply with the toxicological safety parameters outlined by the European Chemicals Agency (ECHA) under the REACH Regulatory Framework. By procuring LASHMAITRE’s certified removal ecosystem, you guarantee that the process revealing the eyelashes after extension removal is executed with absolute, verifiable medical safety.
6. B2B Economics: The Hidden Cost of the “Lash Break”
For the CEO of a vast salon network, the condition of the eyelashes after extension removal must be translated from a biological outcome into a ruthless financial metric. In the professional aesthetic industry, the most devastating financial event is the “Lash Break.”
When a client looks in the mirror and sees damaged, stubby eyelashes after extension removal, they panic. They inform the technician that they need to “take a break” to let their natural lashes heal. To a novice technician, this sounds like a temporary pause. To a B2B corporate economist, the “Lash Break” is the permanent termination of a high-yield recurring revenue stream.
Clients who take a break rarely return. They find alternative cosmetic solutions, or they eventually visit a competitor who utilizes superior, damage-free materials.
Table 1: Financial Modeling – The Catastrophic Cost of the “Lash Break” Churn
| B2B Operational Metric (Per Salon, Per Quarter) | Budget Procurement (Heavy PBT & Toxic Removers) | LASHMAITRE Clinical OEM Procurement (Weightless PBT & Iso-Removers) |
| State of Eyelashes After Extension Removal | Fractured, thinned, suffering from severe Traction Alopecia. | Structurally pristine, biologically healthy, zero breakage. |
| Incidence Rate of the “Lash Break” (Client Churn) | $\approx 25\%$ of clients demand a 3-month break after removal. | $<1.0\%$. Clients confidently transition immediately into a new set. |
| Lost Revenue from Medical Client Churn | 15 lost VIP clients @ $4,000 Annual LTV = **-$60,000** in lost recurring revenue. | $0 lost revenue. Absolute clinical safety secures unbreakable long-term loyalty. |
| Corporate Brand Equity & Regional Dominance | Salon gains a toxic reputation for “ruining natural lashes.” | Salon is revered as an elite clinical authority prioritizing dermal health. |
Industrial Sourcing Insight: Sourcing cheap materials to save $3.00 on a tray or remover is mathematically suicidal if the resulting damage to the eyelashes after extension removal triggers a “Lash Break,” hemorrhaging $60,000 in lost annual Lifetime Value (LTV) per salon location.
7. Cognitive Branding: Monetizing Rehabilitation via OEM Serums
Once a procurement director has secured the highly engineered materials necessary to prevent damage, they must strategically exploit the removal appointment itself. Even when the eyelashes after extension removal are perfectly healthy, the client inherently feels “naked” without their dense synthetic volume. They are psychologically hyper-focused on the length and density of their natural biology.
This psychological vulnerability is the ultimate retail monetization vehicle. The removal appointment should never end with the client simply walking out the door. It must conclude with the mandatory prescription of a clinical-grade eyelash growth serum. Instructing a high-net-worth client to buy a generic serum on Amazon completely destroys the elite clinical authority of your franchise.
This is the exact operational nexus where master academies and regional distributors must aggressively leverage LASHMAITRE’s OEM Private Label capabilities. You are not merely selling a cosmetic liquid; you are providing a proprietary biological catalyst to support the anagen phase of the eyelashes after extension removal.
The LASHMAITRE Premium Visual Aesthetic: Through data-driven analysis of luxury cosmetic consumer psychology, we emphatically advise our B2B partners to embrace a minimalist, text-only logo for their private label eyelash serums. Procurement directors must discard amateurish designs containing literal eye graphics or chaotic colors. A clean, bold, highly sophisticated typographic logo printed on premium matte-white or clinical-black packaging instantly signals dermatological safety, pharmaceutical efficacy, and elite exclusivity. This sleek, text-based aesthetic ensures that your branded serum is perceived as a top-tier medical formulation, effortlessly justifying a $65 to $90 premium retail price point.
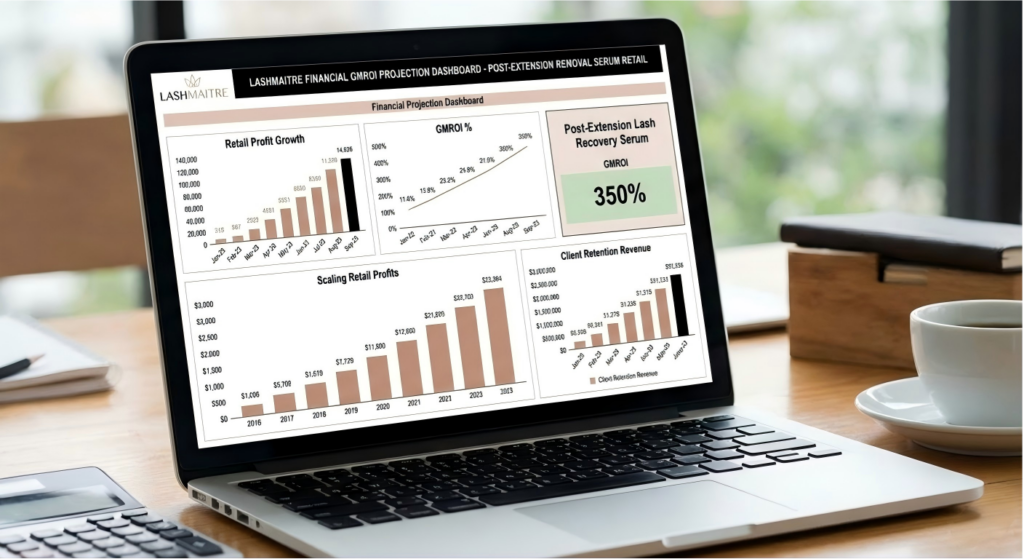
8. Quantitative GMROI: Scaling Retail Revenue Portfolios
The integration of a private label peptide serum targeted at maintaining eyelashes after extension removal represents the highest Gross Margin Return on Investment (GMROI) available in the aesthetic supply chain. Unlike the physical eyelash extensions, which require hours of intensive, highly paid technician labor to generate revenue, the sale of a retail serum requires exactly sixty seconds of consultation time.
By establishing a closed-loop retail ecosystem—where clients are educated that purchasing your proprietary, branded serum is the only way to guarantee massive natural lash growth before their next set—a salon franchise generates massive, passive, recurring revenue that scales infinitely without requiring additional service chairs or technician payroll.
Table 2: Gross Margin Scaling via Private Label OEM Growth Serums
| Retail Operations Metric (Based on a Salon Processing 200 Clients/Month) | Instructing Clients to Buy Generic Drugstore Serums | LASHMAITRE Minimalist Private Label OEM Peptide Serum | B2B Economic Impact |
| B2B Procurement Cost (Per 3ml Retail Unit) | $0.00 | $\approx \$8.50$ (Direct Factory OEM Pricing based on aggregate volume) | Highly accessible procurement cost yielding maximum capital efficiency. |
| Retail Revenue Generated (Selling at $75/Unit) | $0.00 | $\$7,500.00$ (Assuming a highly conservative 50% client conversion rate) | Creates a completely new, high-yield revenue silo independent of labor hours. |
| Clinical Efficacy (Anagen Phase Support) | Unknown risk. Drugstore products may contain harmful prostaglandin analogues. | Maximum Efficacy. Advanced biomimetic peptides formulated strictly to support cellular proliferation safely. | Drastically improves the natural lash baseline, allowing technicians to apply larger mega-volume sets in the future. |
| Monthly Net Retail Profit Added to Salon | $0.00 (Massive Lost Opportunity) | $\approx $6,650.00$ Pure Profit / Month | Generates an additional $\$79,800$ in pure annual profit per salon location. |
9. Advanced FAQ: Technical Sourcing for Procurement Directors
Q: If a client presents with severely damaged eyelashes after extension removal from a competitor salon, how fast can LASHMAITRE’s OEM serum rehabilitate them?
A: Assuming the dermal papilla has not suffered permanent cicatricial (scarring) alopecia from extreme mechanical trauma, the natural follicle requires a full biological cycle to regenerate. LASHMAITRE’s OEM formulations utilize advanced biomimetic peptides (such as Myristoyl Pentapeptide-17) that accelerate cellular proliferation. Visible density and length improvements in the eyelashes after extension removal are typically observed within 4 to 6 weeks of daily application.
Q: Do LASHMAITRE’s OEM growth serums contain prostaglandin analogues that can alter iris pigmentation or cause orbital fat atrophy?
A: Absolutely not. The inclusion of prostaglandin analogues (like bimatoprost) in cosmetic serums introduces severe medical liability, including permanent eye color changes and sunken eyes. Our B2B private label serums strictly utilize safe, FDA-compliant peptide chains and botanical extracts, guaranteeing absolute clinical safety for your brand while powerfully supporting the eyelashes after extension removal.
Q: How does LASHMAITRE handle the logistics of shipping sensitive liquid cosmetics to North America to avoid thermal degradation?
A: We utilize our state-of-the-art Los Angeles B2B distribution node. High-grade peptide serums are highly susceptible to thermal degradation and freezing during traditional ocean freight. LASHMAITRE utilizes priority, climate-controlled air freight to transport bulk liquids to our LA hub, ensuring your private label inventory remains chemically pristine and ready for rapid Just-In-Time (JIT) domestic deployment.
10. Conclusion: Future-Proofing Your Clinical Ecosystem
Treating the condition of the eyelashes after extension removal as a mere byproduct of the industry is a fatal corporate oversight. The health of the natural lash is the ultimate lagging indicator of your entire supply chain’s integrity. Procuring cheap, overweight PBT fibers, highly viscous toxic adhesives, and unregulated chemical solvents mathematically guarantees severe Traction Alopecia, terrified clients demanding a “Lash Break,” and the catastrophic loss of hundreds of thousands of dollars in lifetime value.
By aggressively transitioning your procurement strategy to an engineering and toxicology-focused OEM manufacturer like LASHMAITRE, you entirely insulate your B2B enterprise from these medical and financial liabilities. Our micro-calibrated fibers prevent mechanical trauma, our ISO-compliant removal creams dissolve glue without chemical burns, and our highly lucrative private-label peptide serums transform rehabilitation into a massive retail profit center.
Procurement Directors and Franchise CEOs: Are you ready to completely eradicate the “Lash Break,” mathematically guarantee natural lash health, and absolutely monopolize your regional retail market?
- [Request the LASHMAITRE ISO Toxicology Removal Report and Peptide Serum Sample Kit]
- [Initiate an OEM Private Label Capacity Assessment and LA Logistics Review for Your Network]

Lash Maitre: Your Trusted Partner in Eyelash extension Solutions
Lash Maitre is dedicated to providing professional insights and tips in the eyelash extension industry. Sharing the latest trends, techniques, and product knowledge, Lash Maitre helps lash artists and enthusiasts enhance their skills, stay inspired, and achieve the perfect lash experience.









