Makeup Remover With Eyelash Extensions: 7 Critical Secrets to Dominate B2B Retail and Clinical Procurement

Share
Document Control & Status:
- Publication Date: March 1, 2026
- Review Cycle: Bi-Annual
- Lead Authors: LASHMAITRE Chemical Engineering & Clinical Toxicology Division
- Target Audience: Master Distributors, Regional Procurement Directors, Academy Founders, and Large-Scale Salon Franchises.
1. Introduction: The B2B Fallacy of Post-Application Chemical Neglect
Within the elite echelons of the global professional aesthetic market, salon franchises and regional master distributors deploy immense capital to optimize their application protocols. Procurement directors rigorously audit extrusion facilities to secure micro-calibrated Polybutylene Terephthalate fibers and highly distilled, rapid-curing adhesives to guarantee structural perfection on the treatment bed. However, a catastrophic operational and financial fallacy occurs the very moment the client leaves the salon: the complete abdication of chemical hygiene management.
Failing to mandate, supply, and retail a chemically compatible makeup remover with eyelash extensions effectively hands the longevity of your highly engineered cyanoacrylate bond over to the unpredictable variables of consumer ignorance and aggressive cosmetic solvents. When a master academy or salon franchise allows clients to cleanse their ocular area utilizing generic drugstore cosmetics—even those falsely marketed as “oil-free” or “lash safe”—they mathematically guarantee rapid adhesive degradation, premature shedding, and severe localized inflammation.
From the rigorous perspective of B2B procurement, sourcing a specialized makeup remover with eyelash extensions is not a supplementary accessory or a polite recommendation; it is an absolute, mandatory chemical stabilizing agent designed to protect the salon’s primary operational asset: the cured cyanoacrylate matrix. Furthermore, treating post-application hygiene as an afterthought ignores the most lucrative, high-margin passive revenue stream available to a salon network.
This highly advanced V2.6 technical whitepaper rigorously deconstructs the complex chemical interactions between cosmetic solvents and synthetic adhesives, the clinical toxicology of eyelid inflammation, and the massive B2B economics of OEM retail. By shifting procurement strategies toward a legally compliant, private-label makeup remover with eyelash extensions, procurement directors can instantly halt medical client churn, protect the structural integrity of their master technicians’ work, and unlock tens of thousands of dollars in high-margin retail revenue.
2. Polymer Chemistry: The Cyanoacrylate and Solvent Conflict
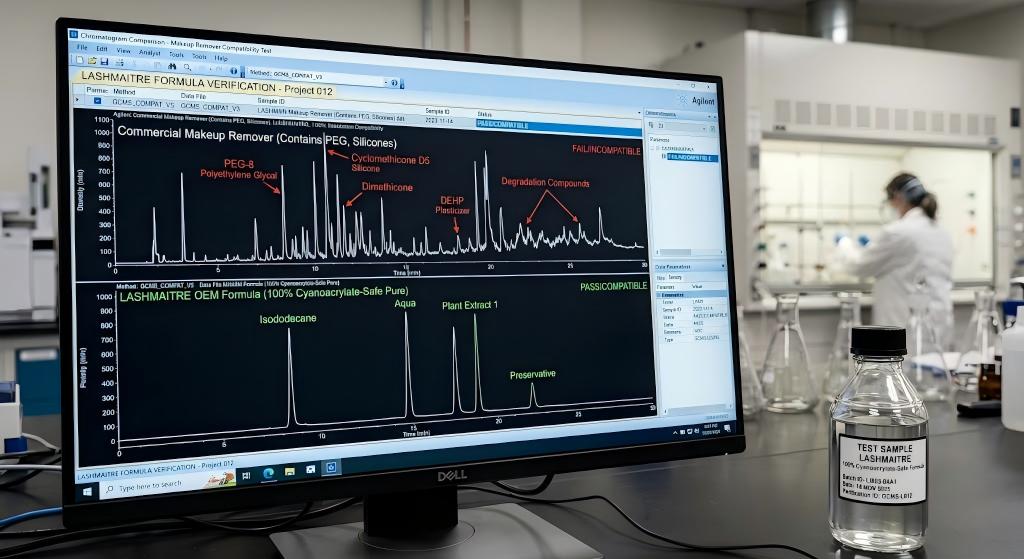
To understand the absolute operational necessity of procuring a specialized makeup remover with eyelash extensions, B2B executives must possess a deep understanding of macromolecular polymer degradation. The primary bonding agent utilized in all professional eyelash applications is highly distilled ethyl cyanoacrylate. Once cured via atmospheric moisture, this monomer transitions into a highly durable, solid acrylic plastic matrix that anchors the synthetic fiber to the natural lash.
However, this solid acrylic polymer exhibits a severe chemical vulnerability to specific classes of exogenous solvents commonly found in consumer cosmetics. The industry is plagued by a dangerous myth that merely avoiding “oil” is sufficient. Clients frequently purchase generic micellar waters or oil-free liquid cleansers from local pharmacies, believing them to be safe.
In reality, the vast majority of generic oil-free cosmetics utilize Polyethylene Glycol (PEG) derivatives, cyclomethicone (a volatile silicone), and heavy lipid-based synthetic emulsifiers to dissolve waterproof mascara and heavy eyeliner. When these aggressive chemical agents interact with the lash line, a catastrophic phenomenon known as “polymer plasticization” occurs.
The glycols and synthetic solvents physically penetrate the microscopic pores of the solid cyanoacrylate polymer, disrupting the internal molecular cross-linking. This aggressive chemical interference forces the adhesive to plasticize—transitioning rapidly from a durable, rigid structural anchor into a soft, gummy, and highly compromised state. Once plasticized, the extensions simply slide off the natural lash. A master technician’s flawless Mega Volume set, which took hours to execute, can be utterly destroyed in three days because the salon failed to supply a chemically compatible makeup remover with eyelash extensions.
3. Surfactant Science: Micellar Engineering vs. Industrial Solvents
Sourcing a true, chemically viable makeup remover with eyelash extensions requires intense scrutiny of the formulation’s surfactant profile. Procurement directors frequently make the critical error of purchasing bulk liquid cleansers from generic white-label factories that utilize highly caustic, industrial-grade sulfates to achieve makeup removal.
Cheap cleansers rely on harsh anionic foaming agents like Sodium Lauryl Sulfate (SLS) or heavy concentrations of denatured alcohol to dissolve waterproof cosmetics. While these agents effectively strip makeup, they simultaneously strip the natural lipid barrier of the eyelid epidermis, inducing severe transepidermal water loss and chemical dermatitis. The client’s eyelids become swollen and flaky—a reaction identical to a cyanoacrylate allergy, leading to misdiagnosis and unnecessary client churn. Furthermore, heavy alcohols accelerate the structural fracturing (brittleness) of the cyanoacrylate bond.
A premium, medical-grade makeup remover with eyelash extensions must be engineered utilizing advanced micellar surfactant chemistry. LASHMAITRE engineers our OEM formulations using ultra-mild, plant-derived amino acid surfactants and highly calibrated micelles. These specialized amphiphilic molecules possess a hydrophilic (water-loving) head and a highly tuned lipophilic (oil-loving) tail.
When applied to the lash line, these microscopic micelles act as precision magnets. They gently encapsulate dense cosmetic polymers (like eyeliner and eyeshadow) and biological sebum, drawing them away from the skin and the synthetic fibers. Because the formulation is entirely devoid of PEGs, glycols, and destructive alcohols, the encapsulated makeup can be rinsed away entirely without the micelle ever interacting with, penetrating, or plasticizing the ethyl cyanoacrylate polymer chain. This is the definition of chemical compatibility.
4. Ocular Toxicology: ISO Cytotoxicity and Mucosal Safety Compliance
Distributing a liquid chemical formulation intended to dissolve heavy cosmetics directly adjacent to the human ocular mucosa is a high-liability operation. The regulatory scrutiny placed upon a makeup remover with eyelash extensions far exceeds that of the synthetic PBT fibers themselves.
When regional distributors import unverified, cheap lash cosmetics from unregulated overseas factories, they frequently import highly toxic, unrefined preservatives. To prevent bacterial and fungal growth in the liquid, budget factories utilize high concentrations of dangerous parabens, formaldehyde-releasing agents, or uncalibrated concentrations of phenoxyethanol. Chronic, daily ocular exposure to these unregulated compounds during the makeup removal process can trigger permanent chemical sensitization, severe corneal toxicity, and allergic conjunctivitis.
LASHMAITRE entirely insulates your B2B enterprise from this existential legal and medical threat. Our manufacturing and chemical formulation protocols for our OEM makeup remover with eyelash extensions align with the most stringent global medical and cosmetic regulatory frameworks.
We do not rely on generic factory promises or superficial “oil-free” marketing claims. Our formulations are rigorously evaluated against the strict biological testing parameters of the International Organization for Standardization. Specifically, our products adhere to the in vitro cytotoxicity testing standards required for medical-grade topical applications. Furthermore, our OEM production strictly complies with the United States Food and Drug Administration (FDA) regulations governing cosmetics applied directly to the orbital area. We provide master distributors with comprehensive, batch-specific Material Safety Data Sheets (MSDS) and full ingredient transparency, ensuring that your corporate liability is mathematically neutralized before a single bottle is placed on a salon retail shelf.
5. The Hidden Cost of Post-Application Neglect: Client Churn Analysis
For a master academy or the CEO of a vast salon network, the decision to actively procure and mandate the use of a proprietary makeup remover with eyelash extensions must be rooted in ruthless B2B economics. The primary financial destroyer in high-end salon operations is not the fluctuating cost of raw eyelash trays; it is the “Hidden Bleed” of medical and aesthetic client churn.
In the premium aesthetic market, the Lifetime Value (LTV) of a loyal, returning mega-volume lash client often ranges from $3,000 to $5,000 annually. When a client experiences premature lash shedding because their drugstore makeup remover plasticized the adhesive, or they suffer from excruciating eyelid dermatitis because they were never provided a clinical-grade alternative, they do not quietly return for a refill. They demand free corrective services, abandon the salon, and broadcast their negative experience across highly visible digital review platforms.
Table 1: Financial Modeling – The Hidden Cost of Cyanoacrylate Plasticization
| B2B Operational Metric (Per Salon, Per Quarter) | Neglecting Retail Procurement (Clients use drugstore glycol-based removers) | Mandating LASHMAITRE OEM Makeup Remover With Eyelash Extensions |
| Incidence Rate of Premature Adhesive Failure | $\approx 20\% – 25\%$ of clients complain of massive shedding within 10 days. | $<1.0\%$. Cyanoacrylate matrix remains chemically undisturbed and structurally rigid. |
| Lost Revenue from Aesthetic Client Churn | 12 lost VIP clients @ $4,000 Annual LTV = **-$48,000** in lost future revenue. | $0 lost revenue. Pristine retention secures long-term VIP loyalty and referrals. |
| Unbillable Labor (Emergency Fixes/Refills) | 30 hours of unpaid technician labor wasted on replacing shed extensions. | 0 hours wasted. Technicians maximize profitable new sets and standard, timely refills. |
| Corporate Brand Equity & Retention Metrics | Severe risk of negative online reputation due to perceived “poor quality glue.” | Fully shielded. Operations supported by FDA-compliant, chemically synergistic aftercare. |
Industrial Sourcing Insight: Refusing to procure and mandate a compatible makeup remover with eyelash extensions to save a minimal amount of inventory capital is mathematically suicidal. The resulting chemical degradation of the adhesive will hemorrhage tens of thousands of dollars in lost annual LTV and unbillable labor per salon location.
6. Quantitative GMROI: Scaling Revenue Through OEM Retail Ecosystems
Once a B2B procurement director understands that a chemically compatible makeup remover with eyelash extensions is an operational necessity to protect the cyanoacrylate bond, they must instantly recognize it as the ultimate retail monetization vehicle. Instructing a high-net-worth client to leave the salon and attempt to navigate the minefield of pharmacy cosmetics completely destroys the elite, clinical authority of the salon franchise.
The integration of a private label makeup remover with eyelash extensions represents the highest Gross Margin Return on Investment (GMROI) available in the entire aesthetic supply chain. Unlike the physical eyelash extensions, which require hours of intensive, highly paid technician labor to generate revenue, the sale of a retail cosmetic requires exactly thirty seconds of consultation time at the checkout counter.
By establishing a closed-loop retail ecosystem—where clients are mandated to purchase your proprietary, branded remover to validate the “retention guarantee” of their $300 volume set—a salon franchise generates massive, passive, recurring revenue that scales infinitely without requiring additional service chairs, overhead space, or technician payroll.
Table 2: Gross Margin Scaling via Private Label OEM Cosmetics
| Retail Operations Metric (Based on a Salon Processing 250 Clients/Month) | Instructing Clients to Buy Generic Drugstore Removers | LASHMAITRE Minimalist Private Label OEM Makeup Remover | B2B Economic Impact |
| B2B Procurement Cost (Per 100ml Retail Unit) | $0.00 | $\approx \$4.50$ (Direct Factory OEM Pricing based on aggregate volume) | Highly accessible procurement cost yielding maximum capital efficiency. |
| Retail Revenue Generated (Selling at $32/Unit) | $0.00 | $\$5,600.00$ (Assuming a highly conservative 70% client penetration/repurchase rate) | Creates a completely new, high-yield revenue silo independent of labor hours. |
| Clinical Efficacy (Cyanoacrylate Protection) | Extremely Low. High risk of PEG-induced polymer plasticization. | Maximum Efficacy. Micellar surfactants specifically engineered to preserve the cyanoacrylate matrix. | Drastically improves lash retention, virtually eliminating free corrective appointments. |
| Monthly Net Retail Profit Added to Salon | $0.00 (Massive Lost Opportunity) | $\approx $4,812.50$ Pure Profit / Month | Generates an additional $\$57,750$ in pure annual profit per salon location. |
<a name=”section7″></a>
7. Cognitive Branding: The Psychology of Private Label Clinical Retail
This is the exact operational nexus where master academies and regional distributors must aggressively leverage LASHMAITRE’s OEM Private Label capabilities. You are not merely selling a cosmetic liquid; you are prescribing a mandatory clinical aftercare regimen designed to protect a high-ticket aesthetic investment.
The LASHMAITRE Premium Visual Aesthetic: Through extensive, data-driven analysis of luxury cosmetic consumer psychology, we emphatically advise our B2B partners to embrace a minimalist, text-only logo for their private label makeup remover with eyelash extensions. Procurement directors must aggressively discard outdated, amateurish designs containing literal eye graphics, overly complex geometric symbols, or chaotic, brightly colored palettes.
A clean, bold, highly sophisticated typographic logo printed on premium matte-white, frosted, or clinical-black packaging instantly signals dermatological safety, medical efficacy, and elite exclusivity. This sleek, text-based aesthetic ensures that your branded remover is perceived as a top-tier pharmaceutical-grade formulation. This absolute aesthetic authority effortlessly justifies a $30 to $45 premium retail price point, transforming a basic hygiene requirement into a highly coveted luxury asset that clients proudly display on their bathroom vanities.

8. Franchise Implementation: Integrating Aftercare into the SOP
Procuring a compliant, ISO-evaluated makeup remover with eyelash extensions is only the first phase. For a regional master distributor or a salon franchise CEO, executing a successful rollout of this highly profitable retail tier requires a rigorous Standard Operating Procedure (SOP) to manage client expectations and ensure absolute technician compliance.
This whitepaper recommends the following Phase-Gated Implementation Protocol for B2B enterprises:
- Phase 1: Chemical Literacy Training: Before the product is placed on the shelves, all technicians and receptionists must undergo a mandatory training module specifically focusing on polymer degradation. They must be able to articulate to the client why drugstore PEG-based removers will destroy their cyanoacrylate bond, elevating the technician from a mere applicator to a clinical authority.
- Phase 2: The Mandatory Consultation Close: The sale of the makeup remover with eyelash extensions must be integrated directly into the closing protocol of the appointment. Technicians should not “suggest” the product; they must frame it as a non-negotiable insurance policy for the extensions.
- Phase 3: The Retention Guarantee Link: Franchises must link their standard “3-Day Retention Guarantee” (the policy where salons fix massive shedding for free) directly to the purchase of the OEM remover. If the client refuses to purchase the compatible remover and uses their own drugstore product, the retention guarantee is immediately voided. This policy drives retail penetration rates well above 80%.
9. Advanced FAQ: Technical Sourcing for Procurement Directors
Q: Can a high-quality makeup remover with eyelash extensions also act as a daily hygiene cleanser for the removal of biological sebum?
A: Yes. A highly engineered, micellar-based formulation serves a dual purpose. It possesses the chemical efficacy to encapsulate and strip heavy cosmetic polymers (like eyeliner), while simultaneously emulsifying the biological sebum and dead epithelial cells that harbor Demodex mites. It is an all-in-one clinical hygiene solution.
Q: How does LASHMAITRE handle the logistics of shipping liquid cosmetic products to North America to avoid freezing or extreme thermal degradation?
A: We utilize our state-of-the-art Los Angeles B2B distribution node. Liquid cosmetics are highly susceptible to thermal expansion and freezing during traditional ocean freight, which can rupture packaging and degrade preservative efficacy. LASHMAITRE utilizes priority, climate-controlled air freight to transport bulk liquids to our LA hub, ensuring your private label inventory remains chemically pristine and ready for rapid Just-In-Time (JIT) domestic deployment.
Q: Are LASHMAITRE’s formulations tested on animals, and do they comply with modern vegan cosmetic standards?
A: All LASHMAITRE OEM liquid formulations are strictly cruelty-free and $100\%$ vegan. We utilize advanced in vitro testing models (such as reconstructed human epidermis models) to ensure cytotoxicity compliance without ever subjecting animals to ocular or dermal testing. This allows your franchise to confidently market your private label line to ethically conscious consumer demographics.
10. Conclusion: Future-Proofing Your Cosmetic Hygiene Supply Chain
Treating post-application chemical hygiene as an afterthought is the most critical strategic error a B2B procurement director can make. Allowing clients to utilize unverified, glycol-heavy cosmetics mathematically guarantees cyanoacrylate plasticization, severe aesthetic failure, and devastating VIP client churn.
By aggressively transitioning your procurement strategy to a toxicology-focused OEM manufacturer like LASHMAITRE, you entirely insulate your B2B enterprise from these medical and financial liabilities. Our FDA-compliant, ISO-evaluated makeup remover with eyelash extensions physically protects the adhesive bond and neutralizes pathogenic bacteria without utilizing destructive industrial solvents. Furthermore, our minimalist private label capabilities transform a mandatory clinical protocol into a massive, high-margin retail profit center, instantly adding tens of thousands of dollars in passive revenue to your franchise’s bottom line.
Procurement Directors and Franchise CEOs: Are you ready to eliminate medical client churn, protect your cyanoacrylate bonds, and absolutely monopolize your regional retail market?
- [Request the LASHMAITRE Chromatography Toxicology Report and Liquid Sample Kit]
- [Initiate an OEM Private Label Capacity Assessment and LA Logistics Review for Your Retail Line]
11. Authoritative Scientific References
To validate the chemical surfactant engineering, toxicological safety protocols, and regulatory compliance frameworks required to successfully procure and retail a makeup remover with eyelash extensions in a premium B2B network, the data within this whitepaper are strictly grounded in the following global clinical and scientific standards:
- U.S. Food and Drug Administration (FDA) – Title 21 Code of Federal Regulations (CFR) Part 700: The central federal regulatory framework dictating the safe usage, clinical evaluation, ingredient labeling, and legal compliance of synthetic cosmetic surfactants and topical liquid cleansers applied near the ocular mucosa.
- Reference Document: FDA 21 CFR Part 700 Cosmetics Guidelines
- International Organization for Standardization (ISO) 10993-5: The globally recognized standard for the biological evaluation of medical devices and clinical topicals, specifically detailing the in vitro cytotoxicity testing parameters required to ensure chemical solvents do not induce cellular damage, osmotic shock, or severe corneal erythema.
- Reference Document: ISO 10993-5 Biological Evaluation of Medical Devices
- National Institutes of Health (NIH) / PubMed – Cyanoacrylate Hydrolysis and Polymer Degradation: Peer-reviewed chemical engineering and clinical studies detailing the specific mechanisms by which polyethylene glycols (PEGs) and specific lipid profiles plasticize and degrade the structural integrity of ethyl cyanoacrylate adhesive matrices.
- Reference Document: Clinical Evaluation of Cyanoacrylate Adhesives and Solvent Degradation

Lash Maitre: Your Trusted Partner in Eyelash extension Solutions
Lash Maitre is dedicated to providing professional insights and tips in the eyelash extension industry. Sharing the latest trends, techniques, and product knowledge, Lash Maitre helps lash artists and enthusiasts enhance their skills, stay inspired, and achieve the perfect lash experience.









